Vertebrate brain theory
for the European Union’s Human Brain Project
ISBN 978-3-00-064888-5
5.9 Motion analysis in the amygdala for maximum coded signals
After the signal divergence in the olfactory cortex had developed, its output was minimum coded. The previous chapter explained how the amygdala converted these signals into maximum-coded signals. Only a signal inversion was necessary. Previously, a switchover to GABA was performed. The now inhibiting signal had an inhibiting effect on the tonic excitation of the inversion neurons, which corresponded to a signal inversion.
The maximum coded signals created a refined image of the environment in the amygdala. They represented scent mixtures.
It was an evolutionary advantage if one could now also see the movement of the associated olfactory objects. For this purpose, however, the amygdala system had to be further developed.
The aim was to transform the maximum coded output signals of the amygdala into a time-delayed and inhibitory signal variant, because this allowed a difference mapping to be generated. For this purpose, this signal variant inhibited the original signals. If nothing had changed in the meantime, both signal components were cancelled. Only the moving objects remained.
The time delay in the vertebrate brain is generally realized by the dopaminergic mean nucleus.
In the first step, the maximum coded output signals of the basal amygdala are sent to the area tegmentalis ventralis where they are switched to dopamine. Their rear projection ends in the central amygdala at GABAergic inversion neurons. These are tonically excited because they receive permanent excitation from the magnocellular basal nucleus. Since these neurons possess the dopamine receptor of type D1, they are inhibited by the dopaminergic input. This corresponds to a first signal inversion. Therefore, these signals are now minimum coded and inhibited, additionally delayed by the long way to the VTA and back.
However, we need maximum-coded signals in the time-delayed and inhibitory variant. So another signal inversion must follow.
This second signal inversion occurs in the medial amygdala. This is structured in the same way as the central amygdala. It also consists of inhibitory neurons that are tonically excited by the mean signal from the basal amygdala. The input from the central amygdala is therefore inverted again. Now the amygdala signals are present with a time delay in the inhibitory and maximum coded variant. They form the inhibitory component of the differential mapping.
The place for this differential imaging is the basal sub-core of the amygdala. Here, in a point-to-point imaging, the original signal of the amygdala in the maximum coded version is combined with the inhibitory, time-delayed and also maximum coded version at the differential neurons. These are only active where there has been movement of the olfactory object in the meantime (the time delay). But also other maximum coded signals (brightness/colours) can be checked in this way with regard to movements.
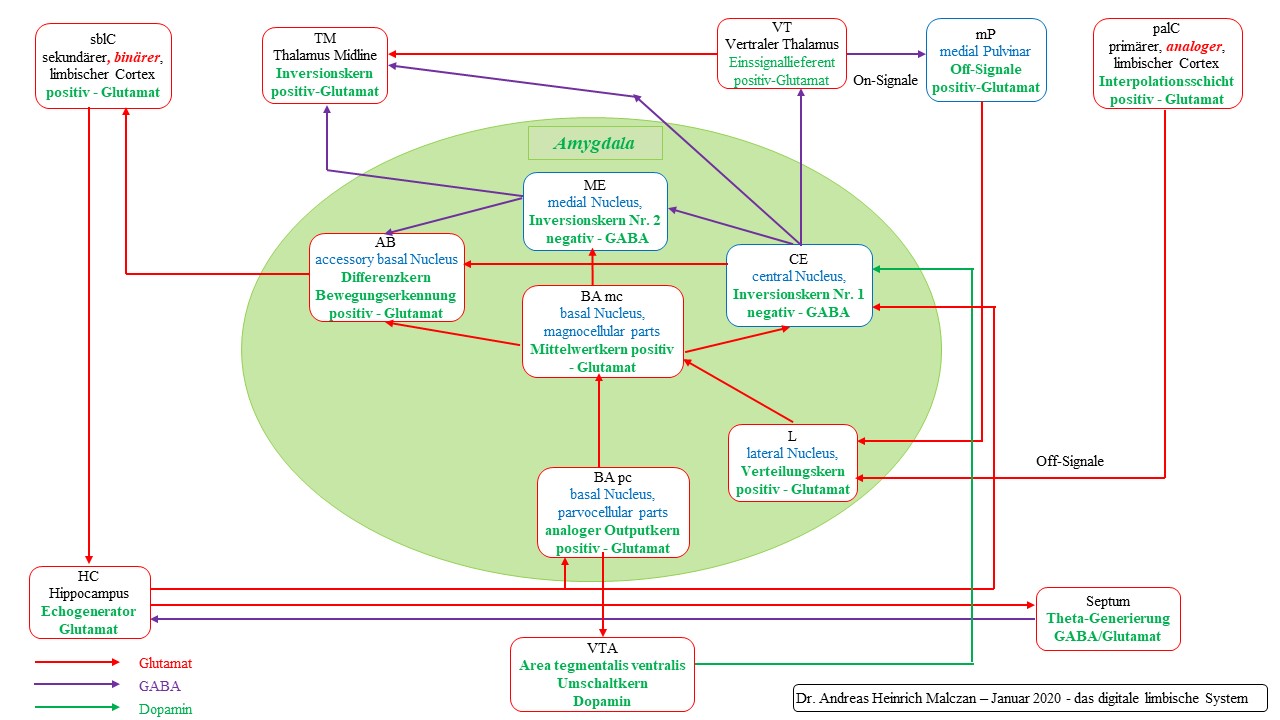
Figure 72 - Basic circuit of the limbic system according to Malczan
Theorem of motion detection in the amygdala for maximum coded signals
Motion detection for maximum coded signals in the amygdala is achieved by connecting a dopaminergic switching core with time delay (VTA) and two inversion cores in series, as well as a differential core in which the time-sensitive difference mapping for motion detection is generated.
The area tegmentalis ventralis (VTA) is the dopaminergic switching core and imposes a time delay on the input.
Both inversion nuclei require a tonic continuous signal for inversion, they obtain it from the mean core of the amygdala, the magnocellular basal nucleus.
Both inversion nuclei consist of GABAergic neurons. The first, the central amygdala nucleus, receives its input to be inverted from the lateral nucleus of the amygdala via the basal amygdala, where the signals contribute to averaging.
The inverted output reaches the medial core of the amygdala where it is inverted again.
The inhibitory and time-delayed signal reaches the basal subcore of the amygdala as the inhibitory component of the differential mapping. The excitatory component is the original signal, which reached this differential nucleus via the lateral amygdala and the basal amygdala.
If the excitatory present signal is stronger than the inhibitory past signal, an excitatory differential signal remains in the differential core, which could trigger reactions.
Thus, the dopaminergic system, consisting of VTA and substantia nigra pars compacta, made motion analysis possible. Of particular importance were the continuous signals, which were indispensable for signal inversion. Failure to provide the permanent signals in the magnocellular basal nucleus of the amygdala or in the subthalamic nucleus deprived the vertebrate of the ability to analyse movement based on short-term memory, which had its roots in the dopaminergic time delay.
It is therefore not surprising that neurologists recommend deep brain stimulation for disorders of the basal ganglia, in which, for example, the subthalamic nucleus is excited with externally generated high-frequency electrical signals that are transmitted to this region via fine probe wires. The external stimulation of the necessary continuous signals in the subthalamic nucleus can alleviate the symptoms that their failure would otherwise cause.
The nucleus basalis Meynert, which functions as a cholinergic mean value nucleus and is located directly next to the globus pallidus, (most likely) supplies the globus pallidus with its mean value signals, which also receives its mean value signals from the nucleus subthalamicus. Therefore, disturbances of the nucleus basalis Meynert also cause a disturbance of the motion analysis.
Theorem of deep brain stimulation
In deep
brain stimulation, an externally generated high-frequency electrical signal
is transmitted via fine probe wires to the mean centre of the basal ganglia
where it can restore the mean signal weakened by a disturbance. This
improves the disease-related symptoms of Alzheimer's and Parkinson's
disease. The signal delivery can take place in the nucleus subthalamicus,
but also in the striatum or globus pallidus. Theoretically, it would also be
possible to apply the signal in the nucleus basalis Meynert. In this way,
affected persons regain the ability to analyse and control movements, but
also improve their cognitive performance, which is made possible by the
change analysis of non-motor signals.
One possible reaction to the time-sensitive differential signal for motion
analysis was the motorized control of the neural clock for propulsion control.
For example, the olfactory difference signal was only present when the animal
swam towards its prey. If an animal happened to move in different directions,
the olfactory difference signal delivered the strongest value when it swam
towards the prey. It could therefore be used directly to trigger these movements
towards the prey. The olfactory difference signal thus resembled the signal of a
radar, it was strongest in the direction of the prey. However, it could only
work when the animal was moving forward. Only then did the strength of the
olfactory gradient increase.
The differential signal could not only be produced in the amygdala system, but also in the nonlimbic system of the basal ganglia. For this purpose, the VTA sent this signal via collaterals simultaneously to the nucleus accumbens, which represented the ventral striatum. There the first signal inversion occurred at the neurons of the shell. These received their tonic signal for inversion from the passing cortical axons. Because of the dopamine receptors D1, these tonic excited neurons were inhibited, so that the input was inverted.
The second signal inversion took place in Globus pallidus, which received its tonic permanent signal for inversion from the nucleus subthalamicus.
The globus pallidus projected the inhibitory component, which was also maximum coded by double inversion, to the thalamus, which also received the original excitatory signals. Thus, a time-sensitive differential image for motion detection was also created in the (ventral) thalamus. The thalamus projected this result to the cortex, where it could be further evaluated.
Thus, the amygdala and the basal ganglia system worked hand in hand and produced the same differential image at different locations.
This is an example of the fact that there are many redundant subsystems in the brain, so that if one system fails, the signals can be evaluated in other "reserve systems".
Monograph of Dr. rer. nat. Andreas Heinrich Malczan